|
Back to Blog
Crystal diffraction energy6/22/2023  
Molybdenum is one of the common target materials for SCXRD. To achieve this, foil or crystal monochromators (e.g. The technique requires K α 1 mainly, which has a slightly shorter wavelength and twice the intensity as K α 2, meaning that K α 2 and K β radiations need to be eliminated. In turn, K α consists of K α 1 and K α 2. Originally, the beam of electrons coming out of a tube mainly consists of K α and K β radiation components. These electrons possess sufficient energy to knock out the inner shell electrons of a specimen giving characteristic X-ray spectra. X-rays are generated in an X-ray tube by heating a filament to produce electrons, accelerating the electrons toward a target by applying a voltage, and impact of the electrons with the target material. Graphical representation of the SCXRD technique (Figure: Nea Möttönen).Īny X-ray diffractometer consists of three basic elements: an X-ray tube, a sample holder, and an X-ray detector. 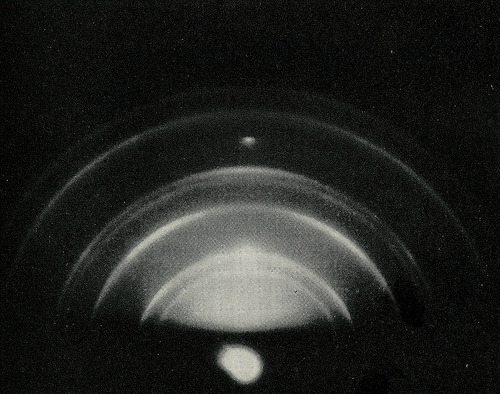
Processing information about the position and intensity of these diffracted X-ray beams (or reflections) yields information about the atomic arrangement within the crystalline material (Fig. The technique is essentially very simple: a single crystal scatters a collimated, monochromatic beam of X-rays, and the scattered beams diffracted by the sample are then measured. In other words, for constructive interference to occur the path difference must be a whole wavelength (nλ where n is integer) giving the Bragg equation for diffraction. The interference is constructive when the phase shift is a multiple of 2π. It was also realized that a Bragg peak occurs only in case when the reflections from various planes undergo the constructive interference. William Lawrence Bragg interpreted this result by presenting a crystal as a set of discrete parallel planes separated by a constant parameter d. They found out that solid crystals, at certain wavelengths and incident angles, produced intense peaks of reflected radiation. The law enabling the identification of the atoms positions has been discovered one year later by W.L. The story of X-ray diffractometry started in 1912, when Max von Laue observed the first X-ray diffraction pattern of the CuSO 4 crystal. This explains why X-rays are used for structure determination. This is the order of bonds and atomic radii. Hence, wavelength used for crystallography is ca. Molecules' size varies from a few to hundreds of ångströms, which is a way too small to be seen with visible light. However, smaller objects require shorter wavelength. Why do we want to use X-ray, not visible light or light of any other wavelength? Visible light radiation falls in range from 400 to 700 nm. X-ray diffraction pattern of a crystallized enzyme. When a specimen is fired with the X-ray beam of incidence, the crystalline atoms cause its diffraction into many directions (Fig.1).įigure 1. Since the diffraction will be similar for adjacent atoms further analysis needs to be done in order to determine the structure of the unknown.Single crystal X-ray diffraction (SCXRD) is a non-destructive technique employed for determination of the atomic structure of a crystal of a certain material (compound). In contrast, X-rays will not give an exact solution if similar characteristics are known between materials. They all have different characteristics, which make neutron diffraction a great technique for identification of materials, which have similar elemental composition. The nucleus of every atom and even from isotopes of the same element is completely different. These lead to a greater and accurately identification of the unknown sample examined if neutron source is being used. These generates several differences between them such as that scattering of X-rays highly depend on the atomic number of the atoms whereas neutrons depend on the properties of the nucleus. Neutrons are scattered by the nucleus of the atoms rather than X-rays, which are scattered by the electrons of the atoms. 
The study of materials by neutron radiation has many advantages against the normally used such as X-rays and electrons. Neutrons have been studied for the determination of crystalline structures. The same relationship is used the only difference being is that instead of using X-rays as the source, neutrons that are ejected and hit the crystal are being examined. \) Bragg’s Law constructionīragg’s Law applies similarly to neutron diffraction. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
